For all studies already IRB approved via Click e-IRB
After the study has been submitted to IRB
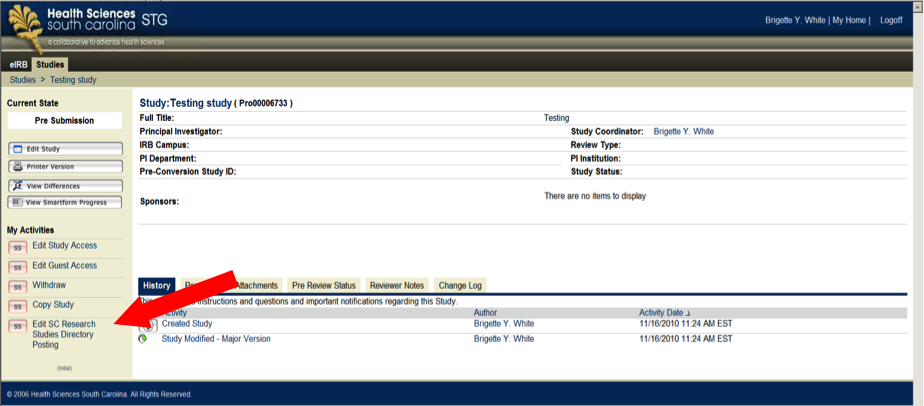
- Select the ‘Edit SC Research Studies Directory Posting’ link under ‘My Activities’ from the protocol’s main page
- In the pop-up box for the ‘SC Research Studies Directory’ Smartform section:
- Ensure the box is checked in the 'Post Study Online' section.
- Enter/Edit Contact Information: Provide the first name, last name, phone number (including area code) and e-mail address of the person you would like the public to contact regarding recruitment for your study.
- Keywords: add/remove all keywords as appropriate to associate with your study.

Note: The lay summary information to be displayed in the directory is taken from the wording entered in the ‘Brief Study Summary’ box located under ‘Study Identification Information’ section within the application. After the study is IRB approved, an IRB amendment must be submitted to edit this language.
Additional Information
You can access your study information at any time to make edits as necessary for sections that do not require an IRB approval (Option to publish, recruitment contact information and Keywords). When your study is no longer approved at the IRB, it will automatically be removed from the public view.
Please note registration of your study on this website does NOT meet the federally mandated requirements of registering with clinicaltrials.gov. To do this, go to http://prsinfo.clinicaltrials.gov





